|
Web Extra Monday, April 16, 2007
T. rex tissue yields genetically revealing proteins
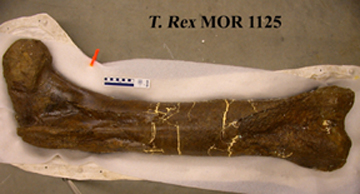 |
| A well-preserved Tyrannosaurus rex femur turned up seven fragments of protein. Three of those fragments matched fragments found in modern chickens, supporting an evolutionary link between dinosaurs and birds. Image is copyright of Science. |
The family tree of an infamous dinosaur is coming to life before researchers' eyes. Scientists say they extracted protein from a 68-million-year-old Tyrannosaurus rex leg bone that supports a genetic link between dinosaurs and birds.
Organic material degrades over time. For example, protein collagen breaks down in skin, leaving human faces with age-revealing smile lines and crow's feet. But exactly how long cells and molecules take to degrade, particularly in fossils, has been debated. Lab and theoretical experiments have suggested that some proteins could last up to 1 million years, "but probably not to the age of dinosaurs," said Mary Schweitzer, a paleobiologist at North Carolina State University at an April 12 press conference. Schweitzer and colleagues' discovery of collagen in a 68-million-year-old dinosaur, described Friday in two papers in Science, confirms that small amounts of such material can survive and perhaps are only now being detected because of increasingly precise molecular analysis technologies.
Friday's description of T. rex protein follows Schweitzer and colleagues' discovery two years ago of the only known well-preserved T. rex soft tissue. The tissue was taken from the same dinosaur, which was excavated in eastern Montana (see Geotimes online, Web Extra, March 25, 2005). Although the team could describe the morphology of the tissue's microstructures, including blood vessels that looked strikingly similar to those of an ostrich, "we could not directly address what the material was made of," Schweitzer said.
To determine how well the tissue was preserved, Schweitzer and colleagues subjected the tissue to a slew of mineral, molecular and chemical analyses, which turned up small, almost undetectable amounts of material that had all the characteristics of collagen — an identifiable and relatively hardy protein. Next, the team turned extracts of the sample over to John Asara, director of the mass spectrometry facility at Beth Israel Deaconess Medical School in Boston. Asara had developed a highly sensitive method using mass spectrometry to sequence protein from various cancer patients to try to identify how the disease spreads to other tissues. The device's sensitivity should be able to determine the sequence of amino acids within the T. rex collagen, the team suggested. If successful, the sequence could then be compared to sequences of modern animals.
The tests worked. Asara found the amino acid sequences for
seven fragments of the collagen protein, three of which most closely matched
those of modern chickens. The remaining sequences matched a mix of other animals,
including a frog and a newt. The finding supports previous evidence that birds
evolved from dinosaurs or that they are closely related, Asara said.
No sequence was unique to T. rex, however, which would have "verified
completely" that the sequence was not modern and thus not the result of
contamination, Schweitzer said. Unique sequences are "like a fingerprint,"
Schweitzer told Geotimes, and also provide more information about an
animal's evolutionary history. The researchers were sucessful, however, in finding
unique sequences from samples from a younger fossil from a mastodon. This fossil
leg bone, which could be as much as 600,000 years old, turned up more than 70
sequences, four of which were unique to the mastodon. Identifying a specific
amino acid at a particular location in the collagen can identify it as belonging
to mastodon.
Derek Briggs, a paleobiologist at Yale University says he is not surprised that the mastodon leg bone had preserved protein sequences, as previous research has already found intact mastodon DNA gene sequences, which are even more delicate. But protein that has survived 68 million years in T. rex is "extraordinary," he says.
Still, Briggs says that the discovery would be more useful if it were known what happens to proteins in other animals in the millions of years between the time of T. rex and the mastodon. "That would demonstrate how potentially powerful [the finding] is, and if it's more widespread than we imagined."
The key lies in getting specimens that are "genuinely
well-preserved," said Jack Horner of Montana State University, speaking
at the press conference. Horner co-authored one of last week's papers on the
tissue and led the team that excavated the T. rex fossils in 2003 from
beneath 1,000 cubic meters of sandstone. "I think we are going to find
that many specimens are like this," he says. "I don't think that this
is unique in any way. I think it's just going to be a matter of paleontologists
taking a little extra time and getting into sites that are not necessarily easy
to excavate."
Links:
"Broken bones
yield T. rex tissue," Geotimes online, Web Extra, March
25, 2005
Back to top

 Subscribe
Subscribe


